Measurement range:
Ca 300 - 498 mg/l (ppm)
Mg 820 - 1800 mg/l (ppm)
About calcium and magnesium:
Along with sodium, potassium, chloride and sulphate, calcium (Ca2+) and mag- nesium (Mg2+) are two key components of natural seawater. Magnesium and,
above all, calcium are important growth factors for organisms which form calcareous skeletons, such as hard coral and coralline algae - both elements form the basic substance of the calcareous skeleton. Furthermore, magnesium and calcium are involved in numerous biochemical processes. In saltwater applications, the reduction in the magnesium or calcium concentration caused partly by cellular metabolism and partly by skeleton formation means it is necessary to test the concentration levels on a regular basis and, if required, add more of one or both ions. This makes it possible to guarantee
optimum, near-natural living conditions for all organisms and to prevent longterm damage. In natural seawater, the calcium concentration is 400 - 410 mg/l (ppm) and the magnesium concentration is 1280-1320 mg/l (ppm). They also have
a fixed ratio of 1: 3.25 to one another. Due to the chemical and biochemical interdependencies between calcium and magnesium, you should also aim to achieve this concentration ratio in saltwater tanks.
Note:
The calcium concentration is determined first, and then the magnesium concentration is determined using the same sample. To avoid cross-contamination, the dosing syringes with the proper tips must only be used again for the same reagents!
Shelf life and storage: 6 months after opening.
Store in a cool, dark place.
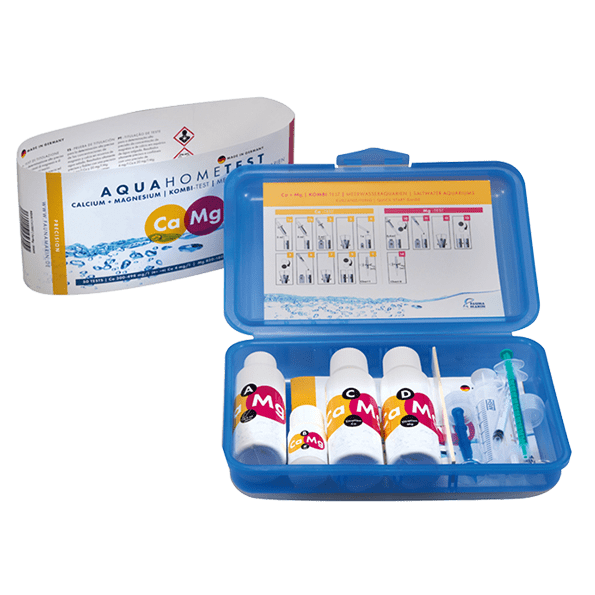
Contents of package:
• 35 ml/1.38 fl.oz. of reagent A
• 9 g/0.32 oz. of reagent B
• 50 ml/1.69 fl.oz. of reagent C
• 40 ml/1.35 fl.oz. of reagent D
• 2 glass cuvettes 10 ml
• 1 dosing syringe 5 ml
• 3 dosing syringes 1 ml with dropper tips
• 1 measuring spoon
• 1 instruction for use