About carbonate hardness / alkalinity:
The carbonate hardness (KH) or alkalinity* of a water sample characterises the buffering capacity, i.e. the ability to maintain the pH value of the water. It is primarily defined by the proportion of hydrogen carbonate ions in the water. As the pH value increases, other basic ions such as the hydroxide ions also contribute to the alkalinity. The alkalinity should be tested in all aquariums regularly. If the alkalinity in the aquarium is too low, the pH value can sink (sudden drop in acidity) to a level which is life-threatening for many fish and invertebrates. In reef aquariums, an adequate alkalinity is essential for strong coral growth. On the other hand, an overly high alkalinity in saltwater tanks can lead to lime precipitates and also have a negative impact on coral growth.
*There are many terms in water chemistry to describe the buffering capacity with varying definitions. The term “carbonate hardness” is customary in reef- and fishkeeping; however, it is the alkalinity which is measured. In this test, both terms are used synonymously. The reading for alkalinity is usually given in degrees of German hardness (°dKH). You can find a table for converting this unit into other common units (such as the equivalence unit millival per litre (mval/l) or the volume of substance in mmol/l) on the second cover page.
Natural seawater has an alkalinity of 6.5 °dKH. The alkalinity in saltwater aquariums should range from 6 to 9 °dKH.
Ask your specialist retailer for the correct alkalinity for your tank.
How to correct unfavourable values:
To increase the alkalinity when levels are too low, we recommend using Fauna Marin Carbonate MIX or Fauna Marin Easy dKH.
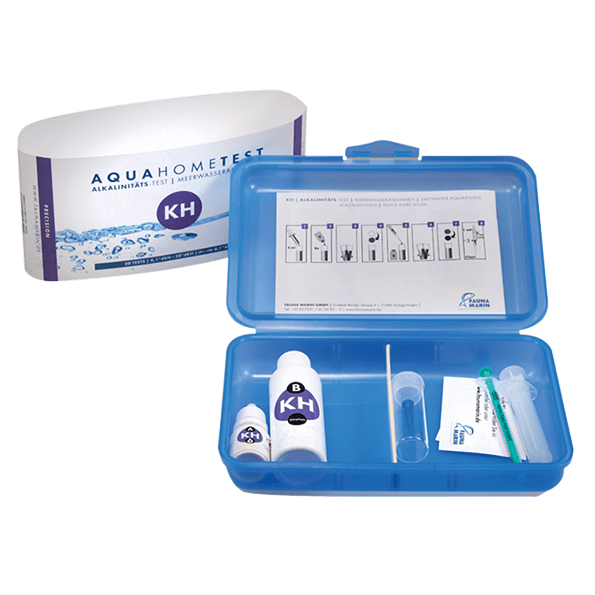
Contents of package:
- 10 ml / 0.34 fl.oz. of reagent A
- 50 ml / 1.69 fl.oz. of reagent B
- 1 glass cuvette 10 ml
- 1 dosing syringe 5 ml
- 1 dosing syringe 1 ml with dropper tip
- 1 instruction for use